
Chemical vapor deposition (CVD) is popular in the semiconductor industry for its ability to produce high-quality, pure, and extremely strong materials. Ultrahigh vacuum CVD (UHV/CVD) requires complex equipment and very high temperatures. To increase efficiency and control costs, engineers can simulate this complex process. Here, we use the growth of silicon wafers as an example.
What Is UHV/CVD?
In CVD, a substrate is exposed to chemical processes that will react or decompose onto its surface to create a deposit, or thin film. We turn to CVD to create high-quality and strong materials, such as graphene, the unique and powerful material that continues to make headlines in the science world. CVD is also valued for its ability to produce a wide range of versatile products, from carbon nanofibers to synthetic diamonds. In semiconductor applications, CVD is used for epitaxial layer growth of high-purity silicon.

Graphene is one type of material that can be formed through UHV/CVD. Image attributed to AlexanderAlUS. Licensed under Creative Commons Attribution-Share Alike 3.0 Unported via Wikimedia Commons.
UHV/CVD is performed at pressures below 10-6 Pa, or about 10-8 torr. In this process, molecular flow is used to achieve gas transport. UHV/CVD doesn’t involve any hydrodynamic effects such as boundary layers and due to the low frequency of molecular collisions, there is no gas-phase chemistry. The growth rate of the material on the substrate is determined by the molecular flux of the species arriving on the surface. Graphene can also be grown using UHV/CVD, as my colleague Daniel Smith wrote about in Part 4 of our blog series on “The Graphene Revolution“.
Simulating UHV/CVD with the COMSOL Multiphysics® Software
As previously mentioned, UHV/CVD experimentation is costly in both time and money, and requires exotic materials and sophisticated thermal management. Fortunately, the Free Molecular Flow interface in the Molecular Flow Module (an add-on to the COMSOL Multiphysics® simulation platform) can simulate this chemical process. For this model, wafers are arranged close together on a movable boat within a quartz tube, surrounded by a furnace. The wafer cassette is positioned within the tube by a delivery rail.
A reactant gas, with a ballast gas, enters the reaction chamber at one end through a load lock. A turbopump is located at the other end of the chamber.
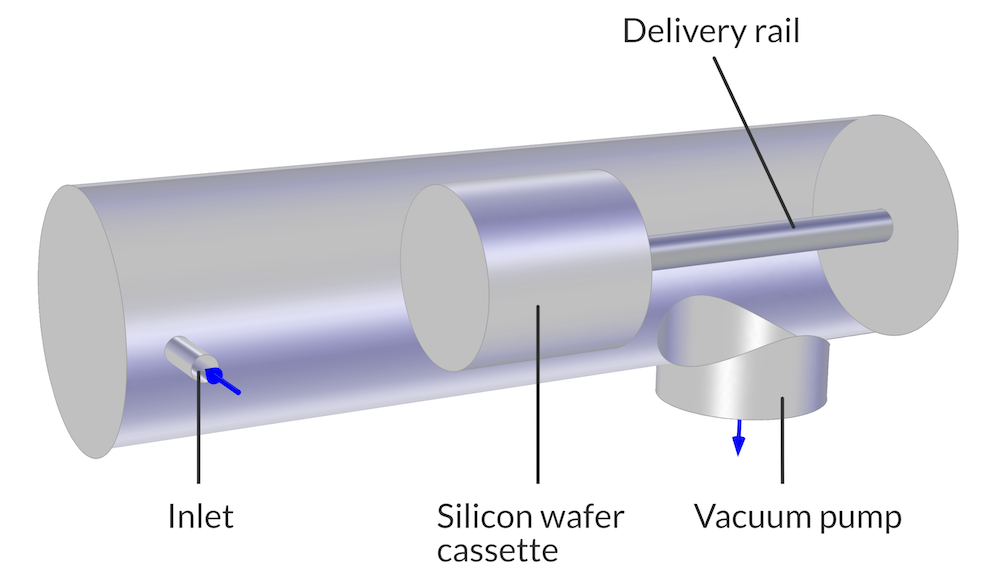
The model geometry of a reaction chamber for a UHV/CVD process.
For this simulation, the reactant gas, silane, enters the inlet of the system with the ballast gas, hydrogen, at a standard mass flow rate of 1 SCCM combined in a ratio of 20% silane and 80% hydrogen. An outgassing wall boundary condition is set for the inlet of the chamber. On the other end of the chamber, a vacuum pump is positioned at a cylindrical port.
Simulation allows us to analyze the process from a range of pumping curves. Three different pumping curves are analyzed for both silane and hydrogen. We enter these curves into the COMSOL Multiphysics software as interpolation functions. Then, we can use a parametric sweep to investigate how the different gases are transported for different pumping curves.
In the surface plot below, we can see the silicon growth on the wafer when using one of the pumping curve options.
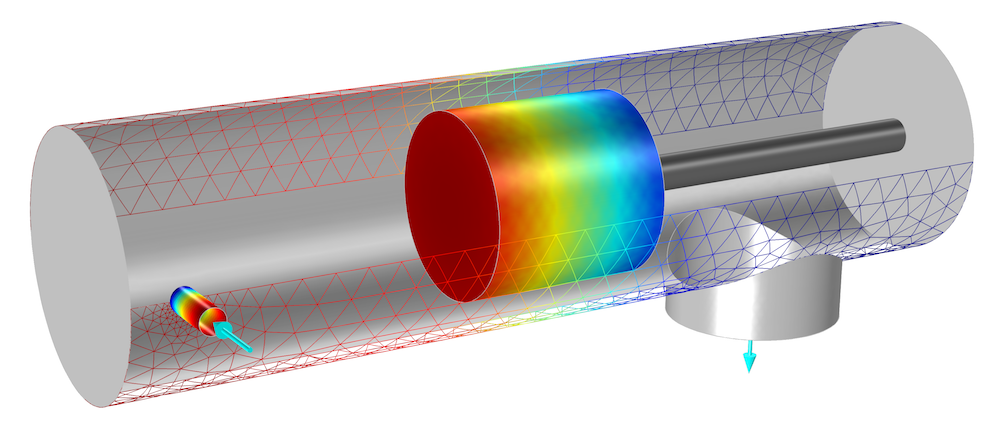
The molecular flux fraction of silane at the wafer cassette, which controls the growth on the wafer.
The molecular flux fraction of silane at the surface of the wafer (0.04) is significantly lower than that at the inlet (0.2). This is because hydrogen is much more difficult to pump than silane, due to its lower molecular weight. Since this measurement directly controls the amount of growth on the wafer, the choice of pump for the silane and hydrogen is important to the amount of material yielded through UHV/CVD. As the molecular flux fraction of each material is hard to measure in a physical experiment, simulation helps to analyze and optimize the UHV/CVD process.
Next Steps
Try this example yourself by downloading the tutorial model from the Application Gallery via the button below.
Further Reading
- Learn more about graphene and how it is formed
- See what else you can model with the COMSOL Multiphysics simulation software
Editor’s note: This post was updated on 5/2/18.




Comments (1)
Agueda Benitez
November 23, 2022Hello! I am tried this example and I am trying to understand each step. I have problems with interpolations. I think there are 6 dataset from 6 real measurements, but I am not sure. Could you explain this section, please?