Design of an Electro-Osmotic Microfluidic Mixer
This paper explores the performance of a microfluidic mixer that uses a sinusoidal electric field to accelerate the diffusion process between two Newtonian liquid species in an electrolytic solution. The 2D model was developed using the Microfluidics module in the COMSOL Multiphysics® software and involved both stationary and time dependent studies. The velocity flow field was investigated, and the relative molar concentration at the outlet was used to quantify how well the two species were mixed.
There are many applications in the chemical, food, and pharmaceutical industries for such devices. In most cases, rapid mixing of different fluid streams is desired. At the small scale of these devices, mixing in the span of a few seconds is desired hence there is an ongoing need for better and more efficient designs.
Microscale mixing devices fall into two classes: (1) passive that rely totally on geometrical stirring or (2) active with moving parts, applied external forces, or an electric field. An active approach was used in this design making use of the electro-osmotic effect. A time-dependent electric field was applied to an array of electrodes within the device to perturb the flow field which would otherwise be highly laminar. The electrodes were placed in strategic interior locations to stimulate the mixing process.
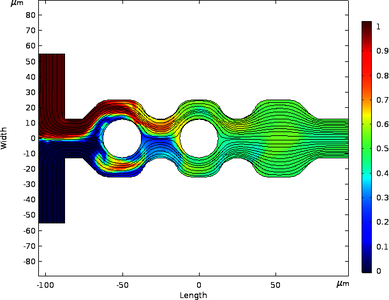
The image below illustrates the final geometry and the resulting streamline velocity field along with the species concentration throughout the device. The design effort focused on two key aspects: geometric shape and the voltage source (location, amplitude, and frequency). The two liquids enter from separate inlets on the left side and exit on the right in a single outlet. Mixing performance is measured by the concentration parameter which ranges from 0 to 1. In those regions with a value near 0.5, the two species are evenly mixed. Both positive and negative electrodes were placed along the wall, each with 0.14 volts of excitation. The time required to reach an equilibrium in concentration at the outlet was 4s.
ダウンロード
- eppes_presentation.pdf - 2.42MB
- eppes_paper.pdf - 0.16MB
- eppes_abstract.pdf - 0.07MB



